ADVERTISEMENT

Medication Safety: A Focus on Pediatric Patients
Part 4 in a 4-part series on medication safety
Our smallest patients are the most vulnerable to medication errors. An awareness of potential vulnerabilities when prescribing in this population is essential.
— Ann M. Dietrich, MD, FAAP, FACEP, Pediatric Emergency Medicine1
Medication safety is critical in pediatric patients because they are especially vulnerable to medication errors. In the two decades since the Institute of Medicine (IOM) released its landmark report, “To Err is Human: Building a Safer Health System,” with recommendations addressing medication errors, the focus on medication safety continues.2 The focus on safety in the pediatric population is essential because most medications used in these patients have never been tested in them.3 Before the U.S. Food and Drug Administration’s (FDA) pediatric program was initiated in 2002, about 20% of all FDA-approved drugs carried pediatric labeling. Between 2002 and 2014, 500 changes in pediatric drug-labeling updates were made,4 with an estimated half of all drugs used in this population containing pediatric labeling.4 Progress continues with labeling changes reaching more than 850 by April 2020.5 For drugs without pediatric labeling, prescribers are using these medications “off-label” based on “sound scientific evidence, expert medical judgment or published literature whenever possible.”6 While many medications have been on the market a long time, without testing and subsequent labeling, there may be unknown factors that can contribute to medication errors and adverse events because drug efficacy, toxicity and pharmacokinetics are different in the pediatric population. This lesson focuses on medication safety in the pediatric population and strategies pharmacists and pharmacy team members can consider implementing to prevent medication errors.
Medication Errors and Pediatric Patients
Twenty-two percent of the U.S. population are under 18 years of age, the pediatric population.7 It is important to use standard age definitions when caring for pediatric patients. See Table 1 for the age associated with preterm, neonate, infant, child and adolescent.8
| Table 1. Pediatric Age Definitions8 |
| Terminology |
Age |
| Preterm |
Birth <37 weeks’ gestation |
| Neonate |
Postnatal age < 1 month |
| Infant |
1–12 months |
| Child |
1–11 years |
| Adolescent |
12–16 years |
Medication errors occur among the pediatric population in many settings, including inpatient, outpatient and in the home environment. The pediatric population has a three times higher risk of medication errors than the adult population in emergency departments, with prescribing errors reported as high as 10%-12%.1 Within pediatric inpatient settings, error rates have been reported between 5%-10%.9 A 2006 systematic review found prescribing error rates between 0.45 to 30.1 errors per 100 orders in the United States and drug administration error rates between 0.6% to 27%.10 Although the analysis was made more difficult because of wide variation in medication error rate definitions and study designs, the magnitude of errors is high. Adverse events and exposures handled by U.S. poison-control centers topped 2.14 million in 2019, an increase of 1.7% over the prior year.11 About half of these exposures were among those ≤19 years old. Nearly 43% of exposures occur in children <6 years old. Therapeutic failures represented nearly 14% of all exposures (294,317) with 40% of these occurring in those ≤19 years old (117,889).11 These therapeutic failures occur most predominantly among children ≤5 years old, followed by those 6-12 years old and those 13-19 years old.11 With regard to site, 67.8% of contacts originated from a residence (own or other) while 92.1% of exposures occurred at a residence (own or other).11
Among the overall population, a recent meta-analysis of studies spanning from 2000 to 2019 suggest one in 20 patients are exposed to preventable harm in medical care with 25% of incidents being medication-related.12 In 2019, there were 5.96 billion prescriptions (30-day equivalent)13 filled in the United States which would result in an estimated 93,600,000 errors given a medication dispensing error rate of 1.57%.14,15
The burden of medication errors is high. Costs of treating drug-related injuries in hospitals are estimated to be $3.5 billion a year,16 and the morbidity and mortality associated with medication errors is estimated to be $77 billion each year.17 The amount of those costs attributable to pediatric patients is not known. Beyond economic costs, errors are costly in terms of patients’ loss of trust, reduced satisfaction and physical and psychological discomfort. They are costly as health professionals lose morale and frustration at providing less than the best care possible.1 Continued efforts to address medication error causes are critical to improve medication safety and public health. Pharmacists and the pharmacy team have important roles to play in preventing medication errors, especially among pediatric patients.
What are Medication Errors?
The National Coordinating Council for Medication Error and Prevention (NCCMERP) defines a medication error as “any preventable event that may cause or lead to inappropriate medication use or patient harm while the medication is in the control of the health care professional, patient, or consumer. Such events may be related to professional practice, health care products, procedures, and systems, including prescribing, order communication, product labeling, packaging, and nomenclature, compounding, dispensing, distribution, administration, education, monitoring, and use."18 Not all medication errors cause harm. When they do, they may be called adverse drug events (ADEs) which are a subset of medication errors and are defined as “an injury resulting from medical intervention related to a drug.”1 See figure one.
Figure 1: Relationship of ADEs and Medication Errors
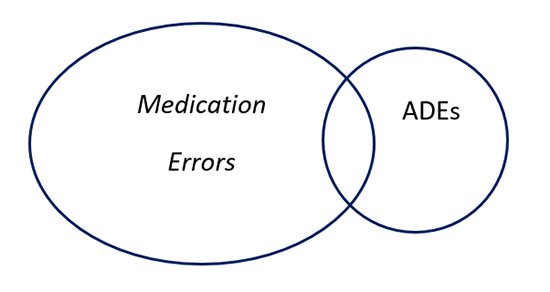
ADEs
Errors may fall into several categories, including errors of omission, commission and system errors.19 Errors of omission are generally when something has not been done right, for example, failure to check patient allergies, weight for proper dosing or provide counseling. Among pediatric patients, these errors may include parents/caregivers not having the correct information to use a medication or not having correct dosing devices to administer medication. Errors of commission are when something has been done wrong, for example, dispensing the incorrect medication, providing the wrong dose or instructions, or if a pediatric patient is given a duplicate dose or a dosing device is used incorrectly providing an unintended dose. System errors are those not exclusively because of an individual’s action, for example environmental factors that distract pharmacy team members during the medication use process or not using technology or processes that can contribute to error prevention (e.g., computerized provider order entry (CPOE), smart pumps, standard order sets). See Table 2 for other examples of each.
| Table 2. Type of Errors19 |
| Definition |
Examples |
| Omission: an error resulting in an inappropriate increased risk of disease-related adverse event(s) resulting from receiving too little treatment (underuse). |
Errors of omission include problems such as failure to include all prescriber instructions, subtherapeutic doses of medications, including the wrong dosing devices or failure to counsel patients/caregivers. |
| Commission: an error resulting in an inappropriate increased risk of iatrogenic adverse event(s) from receiving too much or hazardous treatment (overuse or misuse). |
Errors of commission include problems such as too much medication, treatments that are contraindicated, inadvertently giving the wrong medication, or overriding a medication alert. From a patient perspective they may be repeating a dose, taking expired medications, or storing medication inappropriately. |
| System Error: an error resulting from actions or factors that are part of a process, not just attributable exclusively to an individual’s actions alone. |
System errors include problems such as inadequate lighting, lack of double checks, not double-checking patient names/weights/allergies against orders that result in errors. |
Medication errors can occur throughout the medication-use process. The Institute for Safe Medication Practices (ISMP) is a 501c (3) nonprofit organization devoted entirely to preventing medication errors. In the past 25 years, ISMP has provided tools and resources for healthcare professionals and consumers to help prevent errors. ISMP has defined “Key Elements of the Medication-Use System™20,” noting “Medication use is a complex process that comprises the sub-processes of medication prescribing, order processing, dispensing, administration, and effects monitoring.” Each component may be associated with medication errors as outlined in Table 3. Strategies for preventing these errors have been outlined in the first two parts of this medication safety continuing education series:
- Practical Approaches to Preventing Medication Errors in Community Pharmacy
- Practical Approaches to Preventing Medication Errors in Health System Pharmacy
| Table 3. ISMP’s Key Elements of the Medication-Use System |
| Element |
Error Potential |
| Patient information |
Failure to obtain patient’s demographic (age, weight) and clinical information (allergies) |
| Drug information |
Failure to provide accurate and useable drug information |
| Communication of drug information |
Miscommunication between prescriber, nurse and pharmacist |
| Drug labeling, packaging and nomenclature |
Look-alike, sound-alike names, confusing drug labeling and/or packaging |
| Drug storage, stock, standardization and distribution |
Poor shelf labels, not separating dosage forms on shelves, lack of standardized drug concentrations |
| Drug device acquisition, use and monitoring |
No safety assessments for drug delivery devices or independent double checks, not providing dosage tools/devices |
| Environmental factors |
Poor lighting, noise, interruptions, workload |
| Staff competency and education |
Not focusing on appropriate education, i.e., high-alert medications, policies and processes for medication safety |
| Patient education |
Failure to counsel on medication indications, doses, drug or food interactions, adverse effects, how to protect from errors |
| Quality processes and risk management |
Not analyzing medication error causes and having a system of detecting and preventing errors |
| This table has been adapted with permission from ISMP. https://www.ismp.org/ten-key-elements. |
This lesson’s focus is on understanding the reasons pediatric patients may be prone to experiencing medication-related adverse events and strategies to help prevent them.
Factors Contributing to Medication-Related Problems in Pediatric Patients
Many factors contribute to medication-related problems in pediatric patients, including their size, stage of development and body composition (gastrointestinal [GI], renal, hepatic), inability to communicate about adverse effects and issues with parents, caregivers and providers. Pediatric patients are not small adults. There are developmental, pharmacokinetic and pharmacogenomic differences in the pediatric population which can lead to differences in drug efficacy and toxicity.8 Underdeveloped organ systems impact drug disposition in pediatric patients. Their GI, renal and hepatic systems are different than adults, as is their body composition of water, fat, and plasma proteins.8 Appropriate medication use in pediatric patients must take into account these factors. Table 4 outlines age-related development differences in
| Table 4. Age-Related Development Difference and Impact on Drug Effect8 |
| Characteristic |
Compared to adult |
Age when adult level reached |
Effect |
Example drug(s) |
| Absorption |
|
|
|
|
| pH |
↓ acid production |
2 years |
↑bioavailability of acid-labile drugs
↓bioavailability of acidic drugs |
Penicillin
Phenobarbital |
| GI motility |
↓GI and intestinal motility |
6–8 months |
Longer time to peak concentration |
Digoxin |
| GI content |
↓bile acid & pancreatic enzymes; underdeveloped flora |
~1 year |
↓bioavailability of fat-soluble drugs |
Vitamins A, D, E, K |
| Percutaneous |
↑ absorption |
~4 months |
↑absorption |
Hydrocortisone, rubbing alcohol, salicylic acid |
| Distribution |
|
|
|
|
| Body water |
↑ total body water
↑extracellular body water, rapid change in year 1 |
~12 years |
↑ volume of distribution of water-soluble drugs |
Aminoglycosides, midazolam |
| Plasma proteins |
↓total protein, albumin |
Gradual over year 1 |
↑ volume of distribution and free drug concentration of highly protein bound drugs |
Phenytoin, ampicillin |
| |
Higher free fatty acids |
Gradual over year 1 |
Potential displacement of bilirubin with highly protein bound drugs |
Sulfamethoxazole, ceftriaxone |
| Blood–brain barrier |
Immature, ↓ CNS glycoprotein |
Gradual over year 1 |
↑ CNS penetration |
Aminoglycosides |
| Metabolism |
|
|
|
|
| CYP-450 enzymes |
Lower 1A2, 2C9, 2C19, 2D6, 2E1, 3A4 isoforms |
Adult levels at puberty and varying levels after 4–6 months; varies by isoform |
Altered drug clearance |
Omeprazole, antidepressants, codeine, erythromycin |
| Sulfation |
66%–100% of adult at birth |
Unclear |
|
Acetaminophen |
| N-Acetyltransferase |
Lower |
3–4 years |
Altered drug clearance |
Sulfamethoxazole |
| Excretion |
|
|
|
|
| Glomerular filtration |
Lower, especially preterm |
3–6 months |
↑ elimination half-life |
Aminoglycosides |
| Tubular secretion |
Lower at birth |
7 months–1 year |
↑ elimination half-life |
Penicillins, sulfonamides |
Absorption
Oral drug absorption is impacted by gastric acid secretion, motility and content.8 There is lower acid output at birth until it reaches adult levels at 2 years of age. There is significant change in preterm neonates, at birth and a few days after birth. Acid-labile drugs, such as penicillin, are more bioavailable as a result. GI motility is also decreased, leading to prolonged drug contact with the GI mucosa. This may prolong time to peak concentration yet also result in higher drug levels than those observed in adults for some drugs. Motility reaches adult levels between 6–8 months.8 Infants also have less developed GI contents, including lower bile acids, pancreatic enzymes and underdeveloped flora. This can decrease the bioavailability of fat-soluble drugs, for example vitamins A, D, E and K.
Intramuscular (IM) absorption of drugs is affected by muscle mass, perfusion, stability and muscle contractions and is a consideration in pediatric patients. Percutaneous drug absorption is enhanced in infants because of an immature and more permeable epidermal barrier and a greater body-surface area to weight ratio. This can lead to toxicity with topically administered medications, such as salicylic acid, rubbing alcohol, povidone-iodine and hydrocortisone.8
Distribution
A drug’s distribution is determined by body composition, plasma protein binding, tissue permeation and its chemical properties.8 Total body water decreases and body fat increases with age. Newborns may have 75%–85% body water, for example, compared to 50%–60% in adults. Water-soluble drugs will have greater distribution in pediatric patients while fat-soluble drugs will have lower distribution than in adults.8 On a milligram-per-kilogram basis, neonates require higher doses of water-soluble drugs. Plasma proteins increase with age so highly-protein bound drugs may create toxicity and should be used with caution, e.g., phenytoin, ampicillin, sulfamethoxazole. These drugs may displace bilirubin from protein binding sites causing hyperbilirubinemia and kernicterus, a serious neurological condition.8
Metabolism
The majority of drug metabolism occurs in the liver by the CYP-450 enzyme system. These systems mature at different states of development and drug metabolism is slower in infants compared to older pediatric patients and adults.8 Additionally, glucuronide and oxidation pathways are underdeveloped in infants while the sulfation pathway is well-developed. Acetaminophen is metabolized differently in this age group and they may be at less risk for toxicity although this has not been studied in a clinical trial.8 The glucuronide pathway reaches adult levels by 2 years of age. The maturity of the oxidation pathway varies and is influenced by genetics.8 Genetic polymorphisms can lead to fast or slower metabolism. Codeine and warfarin are examples of drugs that are impacted by this variation.8 The accelerated metabolic process with codeine has been associated with increased morbidity and mortality.21 As a result, the FDA has made label changes indicating that codeine should not be used in those <18 years old.22
Excretion
The renal system is primarily responsible for drug elimination and it matures with age.8 Glomerular filtration, tubular secretion and reabsorption influence the kidney’s excretion of drugs. These processes are lower at birth, with glomerular filtration reaching adult levels within 3–6 months and tubular secretion maturing by 1 year old. As a result, drug clearance is altered with increased elimination half-life in infants.8 Aminoglycoside antibiotics (i.e., gentamicin, tobramycin) will have lower doses in infants because they are eliminated by the kidney; this is to avoid toxic build up and resulting hearing loss and kidney damage.
Besides size and developmental factors, pediatric medication errors may be impacted by the patient’s inability to communicate about a medication’s side effects. Infants do not speak and cannot articulate what they are experiencing. A child’s ability to communicate is influenced by their cognitive development. Parents, caregivers and healthcare providers need to be attuned to signs that there are issues with medications. Pharmacists need to provide education about the child’s medication to parents/caregivers, especially as part of the medication use process. It is important that processes and systems are in place for providing pharmacy services to pediatric patients. The American Society of Health-System Pharmacists (ASHP) and the Pediatric Pharmacy Advocacy Group (PPAG) have developed comprehensive guidelines for providing pediatric pharmacy services in hospitals and health systems which are designed to help pharmacists and pharmacy departments to meet the special needs of pediatric patients.23
Challenges to Pediatric Medication Therapy
While the pediatric population is generally healthier than the adult population, there is still significant medication use in this population. In 2010, more than 263.6 million prescriptions were dispensed to the U.S. pediatric population.24 Analyses have shown a decline in the total number of prescriptions used in the pediatric population between 1999 and 2014 and changes in the most commonly use medications, which vary among infants, children and adolescents.24,25 During 2011-2014, 21.9% of those ≤18 years old had used a prescription medication within a 30-day period. Asthma medications, antibiotics, attention-deficit/hyperactivity disorder (ADHD) medications, topical dermatological/nasal steroid agents and antihistamines are used most often, with amoxicillin the most commonly used drug in infants and children and methylphenidate the top drug dispensed to adolescents.24
A key challenge to using medication in pediatric patients is an incomplete picture of safety, efficacy and pharmacology data in this population, especially among neonates, infants and children under 2 years old.6 This is because most drugs prescribed for pediatric patients have not been tested in them for a variety of reasons historically, ranging from ethical issues such as informed consent to financial issues.3 By necessity, prescribers routinely give medications to pediatric patients "off label,” which indicates the drug has not been FDA-approved for use in children based on the demonstration of safety and efficacy in adequate, well-controlled clinical trials.6
Progress to close gaps in data has been made, stimulated by the passage of two complementary pieces of legislation: the Best Pharmaceuticals for Children Act (BPCA) and the Pediatric Research Equity Act (PREA), enacted in 2002 and 2003, respectively.6 The BPCA provides pharmaceutical manufacturers with an additional 6 months of market exclusivity when clinical trials among pediatric patients are conducted and completed.8 It also allows the FDA to refer drugs to another agency for study if the manufacturer declines to do so. BPCA provides for a systematized review of adverse events in the pediatric population from post-marketing surveillance data through the FDA’s Pediatric Advisory Committee (PAC). If an association is found between a drug and an adverse event, the Agency can request further study of the drug, change the drug labeling to include warnings or contraindications, issue a warning about the drug or even request a drug recall.6 The PREA mandates manufacturers to conduct pediatric studies and create associated labeling if the potential exists for the drug to be used in pediatric patients unless the studies are deferred or waived by the FDA.8 BPCA and PREA have helped incent clinical trials among pediatric patients with more than 800 labeling changes being made since their implementation.3 The FDA maintains a database of these labeling changes—the New Pediatric Labeling Information Database— available at: https://www.accessdata.fda.gov/scripts/sda/sdNavigation.cfm?sd=labelingdatabase.26
For drugs without pediatric labeling, the American Academy of Pediatrics (AAP) has provided recommendations to prescribers on off-label use of drugs in children.6 They note that “absence of labeling for a specific age group or for a specific disorder does not necessarily mean that the drug’s use is improper for that age or disorder. Rather, it only means that the evidence required by law to all inclusion in the label has not been approved by the FDA.” AAP notes that while labeling may be lacking there can be clinical experience and data on a drug’s use in children and that therapeutic decision-making should be guided by the “best available evidence” and the risk/benefit for a patient.6 Peer-reviewed journal articles, AAP practice guidelines and policy statements and handbooks and databases (e.g., Cochrane, Lexicomp and Harriet Lane) are examples of evidence-based resources cited in the guidelines.6 Ultimately, AAP notes, it is the prescribing practitioner who is responsible for deciding what medication and dosage a patient should receive and for what purpose.6 The guidelines also make the point that labeling status should not be the sole criterion for formulary placement, therapeutic substitution or reimbursement.6
Optimizing Pediatric Medication Use
Strategies used to prevent medication errors in pediatric patients can occur during various points in the medication use process, including:
- Patient demographic and clinical information
- Prescribing
- Order communication and transcribing
- Dispensing
- Administering
- Patient education
- Monitoring
Looking at which areas are more frequently associated with medication errors may help to prioritize strategy implementation. An analysis of 428 paid pharmacist liability claims between January 1, 2012 and December 31, 2016 found the most common errors are associated with the wrong drug or dose.27 See Table 5 which compares the distribution of errors to a 2013 analysis which had 164 paid claims among 734 incidents during a 10-year period from January 1, 2002 to December 31, 2011.27
| Table 5: Distribution of Liability Claim Errors27 |
| Claim Error |
Percentage of Occurrence |
| |
2018 |
2013 |
| Wrong drug |
36.8 |
43.8 |
| Wrong dose |
15.3 |
31.5 |
| Contamination of drug/container/equipment |
14.1 |
0.6 |
| Failure to consult with prescriber on question/concern |
5.5 |
4.9 |
| Prescription given to wrong patient |
5.5 |
3.1 |
| Compounding calculation and/or preparation error |
5.0 |
3.7 |
| Failure to obtain/review laboratory values required for proper dosing |
2.8 |
0.0 |
| Labeling error |
2.2 |
0.0 |
| Failure to provide instructions/wrong instructions |
1.7 |
0.0 |
| Failure to supervise |
1.7 |
0.0 |
Several categories saw declines in claims (wrong drug, wrong dose) while others posted gains; notably failure to consult prescribers, failure to review laboratory values needed for proper dosing, giving medication to the wrong patient, and failing to provide instructions or giving the wrong instructions.27
The most frequently occurring causes associated with therapeutic errors reported to Poison Control Centers are similar and outlined in Table 6.11
| Table 6. Therapeutic Error Scenarios from 2019 U.S. Poison Control Center Exposures11 |
| Scenario |
All Cases |
Cases ≤19 years old |
% Among
≤5 years old |
% Among 6-12 years old |
% Among 13-19 years old |
| Inadvertently took/given twice |
93,555 |
30,835 |
16.02 |
11.16 |
5.78 |
| Wrong medication |
47,251 |
15,229 |
15.95 |
10.38 |
5.90 |
| Other incorrect dose |
46,964 |
22,557 |
30.43 |
10.81 |
6.79 |
| Doses too close together |
31,897 |
10,475 |
17.14 |
9.32 |
6.38 |
| Someone else’s medication |
25,917 |
10,310 |
14.84 |
18.18 |
6.76 |
| Other/unknown therapeutic error |
19,308 |
7,005 |
19.27 |
9.87 |
7.14 |
| Incorrect route |
11,345 |
2,083 |
9.19 |
4.41 |
4.76 |
| Confused units of measure |
6,853 |
5,233 |
57.11 |
15.18 |
4.07 |
| Health professional error |
5,913 |
2,345 |
22.14 |
10.59 |
6.93 |
| Wrong formulation/concentration given/taken |
5,551 |
3,899 |
49.54 |
15.73 |
4.97 |
| More than 1 product with same ingredient |
5,050 |
1,863 |
9.84 |
14.26 |
12.79 |
| Dispensing cup error |
3,744 |
3,255 |
67.82 |
15.84 |
3.29 |
| Drug interaction |
3,247 |
642 |
6.25 |
6.56 |
6.96 |
| Wrong formulation/concentration dispensed |
1,619 |
1,105 |
47.56 |
15.38 |
5.31 |
| 10-fold dosing error |
1,302 |
897 |
56.45 |
9.29 |
3.15 |
| Breast-milk exposure |
173 |
156 |
89.60 |
0.00 |
0.58 |
| Adapted from Table 68, reference 11. |
The most common scenarios associated with these therapeutic failures are inadvertently giving/taking medication twice, taking/giving the wrong medication, taking/giving the wrong dose, taking/giving medication too close together and taking/giving someone else’s medication. In the pediatric population, giving or dispensing the incorrect formulation and concentration, errors with dispensing cups during administration and confused units of measure occurred most frequently.11
Consequences of these errors is significant. Table 7 shows the outcomes and costs associated with errors from the liability claims.27
| Table 7: Severity and Cost Associated with Liability Claims27 |
| Error Outcome |
Percentage of Occurrence (%) |
Average Claim Cost Incurred ($) |
| Patient death |
11.3 |
$298,557 |
| Intervention to save patient’s life |
2.8 |
291,615 |
| Permanent patient harm |
17.4 |
274,873 |
| Error reached patient, no harm |
1.6 |
102,833 |
| Temporary patient harm requiring intervention/prolonged hospitalization |
41.3 |
72,577 |
| No medication error |
1.1 |
28,750 |
| Patient monitoring required to confirm no harm or intervention needed |
1.1 |
19,322 |
| Temporary patient harm requiring intervention |
23.4 |
10,387 |
A look at factors associated with the wrong drug dispensing liability claims reflect failures in one or more of the key elements of the medication use system (refer to Table 2.)27 These include:
- storage and quality process issues related to look-alike, sound-alike drugs;
- failure to check drug against label and actual prescription;
- failure to consider patient history/medication profile/drug therapies;
- failure to review the prescription with the patient; and,
- failure to check with the prescriber.
Addressing these factors is important in preventing medication issues among the pediatric population.
Patient Demographic and Clinical Information
Steps should be taken at every encounter to ensure a patient’s profile has all current demographic and clinical information, including the patient’s weight in the metric system, laboratory values, medical conditions, medications (prescription, OTCs and supplements) and immunizations. Most pediatric medications are dosed according to body weight, some are dosed by body surface area (BSA).8 For pediatric patients, weight and BSA changes over time. Having up-to-date information is critical to double-checking medication orders. It is a medication safety best practice to be sure pediatric patients are weighed using metric units of measure and that these weights are documented in the medical record and on the prescription.28
Making note of parents and any additional caregivers and their contact information can help when changes occur to a patient’s regimen and new information needs to be given to the patient’s care team. The profile should also have the patient’s primary and specialty providers so questions can be directed to the appropriate clinician as well as any recommendations related to the patient’s medication regimen. Having accurate demographic and clinical information provides the baseline needed to evaluate any new prescription order(s) for the patient or when conducting a medication intervention such as medication reconciliation or a chart review.
Prescribing Errors and Appropriate Prescribing
Prescribing errors contribute to medication errors and occur frequently in pediatric patients as noted earlier. Prescribing errors may be associated with:
- Illegibility
- Inappropriate abbreviation
- Directions
- Strength
- Dose
- Length of treatment
- Frequency
- Amount
Many errors can be eliminated through the use of e-prescribing with clinical decision support.29 A systematic review of interventions used to reduce pediatric medication errors reported a 36%–87% reduction in prescribing errors with CPOE with clinical decision support and 27%–82% with standardized preprinted order sheets.29 Besides decreasing prescribing errors, e-prescribing has been shown to increase efficiency, decrease patient abandonment of prescriptions and help save on healthcare costs.30,31 While electronic prescribing has many benefits, errors within the process can still occur. The pharmacy team needs to be vigilant to carefully verify the patient’s information, drug, dosage, instructions, and physician, entering notes into the fill or patient records to clarify any issues with the order to ensure continuity of care with all pharmacy team members.
Best practices for prescribing to prevent pediatric medication errors include:28
- Ensuring patient’s weight in kg or g is entered in the CPOE system and medical record prior to orders being entered;
- Enabling dose range checking software within CPOE, e-prescribing systems and pharmacy computer systems;
- Checking for drug allergies, interactions and contraindications prior to prescribing and making appropriate notes in the medical record;
- Including both the mg/kg or mg/m2 dose and the calculated amount per dose for pediatric drug orders;
- Generally, pediatric doses should not exceed the recommended adult dose;
- Ordering pediatric liquid medications in metric doses, not volume only;
- Ordering each ingredient of a parental nutrition(PN)/complex electrolyte solutions as dose/kg/day (e.g., mg/kg/day, mcg/kg/day) for younger children; and,
- Ordering each ingredient of a parental nutrition(PN)/complex electrolyte solutions per day for older children.
There will still be times when prescriptions are called in verbally, presented manually in written form or sent to the pharmacy via fax. There are strategies that the pharmacy team can employ to help reduce the possibility of errors, including reviewing each prescription to be sure it includes the patient name, age, metric weight, drug name, dosage form, strength, dose frequency and route, indication, prescriber information and the person transmitting the order.32 Consider using templates or prescription pads containing all elements of an ideal order. Always read back the prescription to the person giving the order when taking verbal orders. Transcribe the order neatly, print is preferable, using the metric system. Include leading zeros, used before a decimal quantity less than one, e.g. write 0.1 not .1. Trailing zeros should NOT be used after a decimal. e.g., write 1 not 1.0. Finally, do not use abbreviations when writing prescriptions. The Joint Commission has a list of abbreviations that should never be used, see Table 8.33
| Table 8. The Joint Commission Official “Do Not Use” List32 |
| Do Not Use |
Potential Problem |
Use Instead |
| U (unit) |
Mistaken for “0” (zero), the
number “4” (four) or “cc” |
Write “unit” |
| IU (international unit) |
Mistaken for IV (intravenous)
or the number 10 (ten) |
Write “international unit” |
| Q.D., QD, q.d., qd (daily) |
Mistaken for each other
Period after the Q mistaken for
"I" and the "O" mistaken for "I" |
Write “daily” |
| W.O.D., QOD, q.o.d., qod (every other day) |
Mistaken for each other
Period after the Q mistaken for
"I" and the "O" mistaken for "I" |
Write “every other day” |
Trailing zero (X.0 mg)
Lack of leading zero (.X mg) |
Decimal point is missed |
Write X mg
Write 0.X mg |
| MS |
Can mean morphine sulfate or
magnesium sulfate
Confused for one another |
Write “morphine sulfate” |
| MSO4 and MgSO4 |
Can mean morphine sulfate or
magnesium sulfate
Confused for one another |
Write “magnesium sulfate” |
| Additional Abbreviations for possible future inclusion |
| Do Not Use |
Potential Problem |
Use Instead |
>(greater than)
<(less than) |
Misinterpreted as the number
“7” (seven) or the letter “L”
Confused for one another |
Write “greater than”
Write “less than” |
| Abbreviations for drug names |
Misinterpreted due to similar
abbreviations for
multiple drugs |
Write drug names in full |
| Apothecary units |
Unfamiliar to many
practitioners
Confused with metric units |
Use metric units |
| @ |
Mistaken for the number
“2” (two) |
Write “at” |
| cc |
Mistaken for U (units) when
poorly written |
Write “mL” or “ml” or “milliliters” |
| µg |
Mistaken for mg (milligrams)
resulting in one thousand-fold
overdose |
Write “mcg” or “micrograms” |
In pediatric patients, it is especially important to be sure prescribed medication is appropriate, that the dosage has been adjusted based upon the patient’s metric weight and stage of development. Use of the indication, effectiveness, safety and adherence medication assessment approach which is part of comprehensive medication management patient care process is a useful tool outlined in Table 9.34 During transitions of care, medication reconciliation is an important tool. During the reconciliation process all medications currently being taken by the patient are verified, with each drug reviewed for appropriateness for the situation/setting, reconciling the new drug list with the previous list with all changes and reasons for changes documented and communicating the updated list to the next care provider.35
| Table 9. Medication Assessment Process34 |
| 1 |
Is there an indication for the drug? |
| 2 |
Is the medication appropriate for the medical condition being treated? |
| 3 |
Is the dosage, frequency and duration correct? |
| 4 |
Are the directions correct and practical? |
| 5 |
Are there clinically significant drug-drug interactions? |
| 6 |
Are there clinically significant drug-disease interactions? |
| 7 |
Are there clinically significant drug-food interactions? |
| 8 |
Is there unnecessary duplication with other drugs? |
| 9 |
Is the patient experiencing any side effects? |
| 10 |
Is this drug the most affordable option to optimize adherence? |
Dispensing
Numerous strategies exist to help reduce dispensing errors. A primary contributor to wrong drug errors is failure to take special precautions with sound-alike and look-alike drugs (15.1%).27 These medications should be separated on pharmacy shelves using color or tall man letters. The ISMP maintains a list of look-alike drug names with tall man letters.36 Examples of the list may be found in Table 10.
| Table 10. Examples of Look-Alike Drug Names with Tall Man Letters |
| Drug Name with Tall Man Letters |
Confused With |
| diphenhydrAMINE |
dimenhyDRINATE |
| FLUoxetine |
DULoxetine – PARoxetine |
| oxyMORphone |
HYDROmorphone – oxyCODONE – OxyCONTIN |
| prednisoLONE |
predniSONE |
| risperiDONE |
rOPINIRole |
| This table has been adapted with permission from ISMP. https://www.ismp.org/sites/default/files/attachments/2017-11/tallmanletters.pdf |
Among pediatric patients, examples of paid claims involving wrong drug include27:
- Methadone 10 mg dispensed instead of methylphenidate 10 mg resulting in severe fatigue requiring emergency treatment
- Doxepin 100 mg dispensed instead of doxycycline 100 mg resulting in altered mental status, dizziness, pallor and insomnia requiring hospitalization.
- Glyburide dispensed instead of clonidine resulting in hypoglycemic crisis requiring hospitalization.
Some of these errors involved high-alert medications. High-alert medications are drugs that bear a heightened risk of causing significant patient harm when they are used in error.37 ISMP maintains lists of high-alert medications for both the community/ambulatory and acute care settings. Pediatric liquid medications that require measuring are on the list. Most institutions maintain a list of pediatric high-alert medications and standardize/limit the concentrations and strengths available.28 Chemotherapeutic drugs, immunosuppressive medications, lipid/total PN and opioids are among high-alert medication classes for inpatient pediatric patients generated through an international modified Delphi study.38 Specific medications include amiodarone, digoxin, dopamine, epinephrine, fentanyl, gentamycin, heparin, insulin, morphine, norepinephrine, phenytoin, potassium, propofol and tacrolimus.38
High-alert medications should be flagged with additional care taken to ensure the correct medication has been selected with the correct dosage. ISMP suggests using auxiliary labels, shelf labels and automated alerts and standardizing the storage, dispensing and administration of these products.37
Best practices for preventing dispensing errors include having double-checks built into the workflow systems, including use of automated compounding devices for PN/complex electrolyte solutions, verification of the mg/kg or mg/m2 dose used to calculate the final dose on the prescription order before preparing/dispensing the medication, ensuring the dose-checking software is enabled in the pharmacy computer system, barcode verification systems, and having a clinical pharmacist present on patient care units to provide input during prescribing/dispensing.28
When dispensing medications for pediatric patients, appropriate auxiliary labels should always be included as a reminder for parents/caregivers (e.g., shake well, refrigerate, take with food, avoid sunlight). Providing flavoring for liquid medications is a common service for pediatric patients. Proper dosing devices using the metric system should be included for dosing liquid medications, such as calibrated measuring cups, droppers and oral syringes. The devices included in packaging for over-the-counter medications should be used for those medications.
Administration
Addressing issues in medication administration is an important strategy to prevent medication errors since pediatric patients often depend on others to give medications. In the institutional setting, double verification of the patient, drug, dose, line attachment, pump settings and infusion rates should be conducted.28 Another best practice is the use of barcode scanning at the patient bedside. Smart infusion pumps with activated drug libraries should be used for pediatric drug administration.28 See the second lesson in this continuing education series for more detail–Medication Safety: Practical Approaches to Preventing Medication Errors in Health System Pharmacy.
In all settings, appropriate dosing devices using metric measurement should be dispensed with medications. Patients and/or parents/caregivers need to understand how to use these devices correctly. Numerous studies have documented issues with parental and caregiver use of dosing devices.39 One study evaluated the accuracy of parents’ use of etched calibration dosing cups, printed calibration dosing cups, dropper, dosing spoon and oral syringes with and without bottle adaptors.40 The lowest accuracy was associated with dosing cups with etched markings (30.5% accuracy) and those with printed markings (50.2% accuracy). Researchers found dosing cups were associated with increased odds of dosing errors compared with an oral syringe.40 More than 85% dosed accurately with the remaining instruments.40
The FDA issued voluntary industry guidelines that recommend greater consistency and clarity in pediatric OTC medication dosing directions and their accompanying measuring devices in 2011.41 This guidance has driven change and most OTC pediatric liquid medications adhere to the FDA recommendations for dosing directions and devices.42
Patient Education and Monitoring
Patient education is an important double check in the medication use process and gives the patient and/or parent/caregiver needed information to use the medication correctly. For younger patients and those who have limited cognitive development, education efforts may need to be directed to the parent/caregiver. For older pediatric patients and adolescents counseling should be age-appropriate. Before beginning a counseling session, identify any potential barriers, such as literacy issues, vision, hearing or speech problems, stigmas, or any other concerns. The counseling should be adjusted as different barriers are discovered. Review the drug’s name, purpose, appearance, dose, route (chew, apply, crush, etc.), administration schedule, side effects and precautions, and proper storage, with written instructions provided. The use of pictograms in addition to text-only written information should be considered since they have been shown to reduce the risk of medication dosing errors.43 Specific instructions should be given to the patient and/or parent/caregiver on how to use the dosage device dispensed with the medication and the difference between milliliters and teaspoons for liquid medications. Patients and/or parents/caregivers should also be advised not to share medications between siblings. The pharmacist should reinforce the importance of reading the medication label, including for any OTC medications, and to avoid duplicating ingredients. If a different manufacturer is used and the medication’s size, shape and/or color has changed, it is important to bring this to the parent’s/patient’s attention.
Proper storage of both prescription and OTC medication is important to avoid medication errors and accidental ingestion.44 Medications and vitamins should be stored out of the reach and sight of children. Secure medication storage containers and individual vials are commercially available. Placing medications in a locked drawer or on a high closet shelf are alternative storage sites. Children should be taught that medicines are not candy and that they should not touch, sniff or taste them on their own. Parents/caregivers should have contact information for the closest Poison Control Center or call 800.222.1222 in the event of an accidental ingestion.
Ask the patient and/or parent/caregiver what questions they have and provide appropriate follow up. Use the teach-back method and have them repeat what the medication is for, how they will take/give it, what side effects to look for and what to do if a dose is missed. Using the three-question technique for new and refill medications developed by the Indian Health Service may be an efficient approach to providing counseling.45 See to Table 11
| Table 11. Indian Health Service Three Prime Questions Technique for New and Refill Prescriptions45 |
| Questions New Prescriptions |
Content Provided |
| What did the prescriber tell you the medication was for? |
Name and purpose of the medication |
| How did the prescriber tell you to take the medication? |
Dose, route, frequency, storage, duration, and use techniques |
| What did the prescriber tell you to expect? |
Positive outcomes expected, what to do if they do not occur; possible side effects, how to decrease occurrence and actions to take if they do occur |
| Questions Refill Prescriptions |
Content Provided |
| What do you take this medication for? |
Purpose of the medication |
| How do you take it? |
Directions and techniques for use |
| What kind of problems are you having? |
Perceived side effects |
Patients and/or parents/caregivers should be encouraged to always double check their medications and call their pharmacist if there is anything unusual. Providing them with access to their child’s medication profile can be helpful. Encourage parents/caregivers to keep their child’s current medication list to share with all their care providers.
Medication monitoring can occur with each encounter. Another method of monitoring is post-dispensing follow up phone calls to patients and/or parents/caregivers to see if they have questions. Ask if the patient is experiencing any issues and follow up as needed. Ask if they have been able to administer the medication as intended and adhere to the prescribed regimen. Medication nonadherence can lead to other problems including disease progression, treatment failure, hospitalization and ADRs. Counseling can also reinforce medication adherence. Medication synchronization, automated refills and refill reminder services can support adherence, especially for pediatric patients on therapy for chronic conditions.
Resources for Parents and Caregivers About Medication Use in Pediatric Patients
There are many resources geared to parents and caregivers designed to help them use medications correctly in pediatric patients and to report medication-related issues. The pharmacist and pharmacy team members should be familiar with these resources and recommend them when appropriate.
The ISMP operates the ConsumerMedSafety website (https://consumermedsafety.org/). The website has medication safety articles on a variety of topics, including “Keeping Children Safe. ” There are numerous articles within this topic, including but not limited to:
- protecting children from medication mishaps;
- ensuring medications are used safely at schools and daycare;
- keeping medication stored safely to avoid accidental ingestion;
- antibiotic overuse; and,
- cough/cold medication use in pediatric patients.
Parents and caregivers can sign up for complimentary newsletters and access tips and tools for using medications safely. They can also see FDA safety alerts and report any problems or medication errors to the ISMP’s National Medication Errors Reporting Program (ISMP MERP).46 The ISMP MERP was established in 1975 and is a voluntary medication error-reporting program (https://www.ismp.org/report-medication-error). The program’s objectives are to:
- Learn the underlying causes of reported medication errors or hazards
- Disseminate valuable recommendations to organizations to prevent future errors
- Provide guidance to healthcare community, regulatory agencies, and pharmaceutical and device manufacturers
Another resource is the National Council on Patient Information’s (NCPIE) BeMedWise.org website (www.bemedwise.org). NCPIE is one of the original patient safety coalitions. The website is a tool for consumers, patients, caregivers and healthcare professionals alike. One section is designed for parents and caregivers to help them use medications safely in children (https://www.bemedwise.org/how-and-when-to-use-medicines-for-children/). There is also a toolkit for parents, community leaders and health educators with these tip sheets:
- Top 10 medicine use tips for parents and families;
- Ask the right questions: Questions to ask about the medicines you and your family members take;
- FamilyWize: A guide to smart, safe prescription use and savings;
- What’s your family’s medicine action plan? Medicine safety for families; and,
- The Dos and Don’t of Medicine Disposal in Your Home.
The toolkit can be accessed at: https://www.bemedwise.org/health-research-and-reports/health-resources-and-toolkits/toolkit-for-parents-community-leader-and-health-educators/.
The FDA also has a wide variety of children’s health information available at: https://www.fda.gov/consumers/consumer-updates/childrens-health. Examples of relevant medication-related topics include:
- Medication safety: advice for new parents;
- Allergy relief for your child;
- When to give kids medicine for coughs and colds;
- Dealing with ADHD: what you need to know;
- Know active ingredients in children’s meds;
- Don’t double up on acetaminophen; and
- Lock it up: medication safety in your home.
The Centers for Disease Control and Prevention (CDC) also has information to help parents and caregivers use medication safely in their children available at: https://www.cdc.gov/patientsafety/features/safe-medicine-children.html. Pharmacists may wish to refer patients/caregivers to these resources or adapt some of the resources to make them pharmacy-specific, especially for new patients.
Summary
Everyone on the pharmacy team has a role to play in preventing medication errors, especially among pediatric patients. Pediatric patients have physical and cognitive development that requires medications be dosed differently compared to adults. Medications can greatly improve health when used wisely and correctly. Providing counseling to parents and caregivers so they are able to properly administer medications to children is critical. Medication errors do occur and cause preventable human suffering and financial cost. There are a number of strategies that can help the pharmacist and pharmacy team members prevent medication errors among pediatric patients and optimize their medication therapy. Being aware of issues facing pediatric patients and following best practices to reduce medication errors can contribute to making medication use safe for your pediatric patients.
References
- Morizio K, Edwards CJ, Jarrell DH. Pediatric medication safety. November 1, 2018. Reliasmedia.com website. Available at: https://www.reliasmedia.com/articles/143437-pediatric-medication-safety. Accessed December 28, 2020.
- Kohn LT, Corrigan JM, Donaldson MD, Ed. To Err is Human: Building a Safer Health System. Institute of Medicine: Committee on Quality of Health Care in America. Washington D.C.: National Academy of Sciences. Available at: http://nap.edu/9728. Accessed December 28, 2020.
- Drug research and children. U.S. Food and Drug Administration website. May 4, 2016. Available at: https://www.fda.gov/drugs/information-consumers-and-patients-drugs/drug-research-and-children. Accessed December 28, 2020.
- Haelle T. 500 drugs updated with directions for child use since 2002. March 10, 2014. ScientificAmerican.com website. Available at: https://www.scientificamerican.com/article/500-drugs-updated-with-directions-for-child-use-since-2002/. Accessed December 28, 2020.
- New pediatric information labeling database. U.S. Food and Drug Administration website. August 31, 2020. Available at: https://www.accessdata.fda.gov/scripts/sda/sdNavigation.cfm?sd=labelingdatabase. Accessed December 28, 2020.
- American Association of Pediatrics Policy Statement. Off-label use of drugs in children. Committee on Drugs. Pediatrics 2014;133:3:563-567. Available at: https://pediatrics.aappublications.org/content/133/3/563. Accessed December 28, 2020.
- Total population by child and adult populations in the U.S. The Annie E. Casey Foundation Kid Count Data Center. Available at: https://datacenter.kidscount.org/data/tables/99-total-population-by-child-and-adult-populations#detailed/1/any/false/1729,37,871,870,573,869,36,868,867,133/39,40,41/416,417. Accessed January 9, 2021.
- Poon CY, Lee CKK. Pediatric pharmacy practice. In Remington: The Science and Practice of Pharmacy. 22nd ed. Philadelphia, PA: Pharmaceutical Press; 2013.
- Benjamin L, Frush K, Shaw K, et al. Pediatric medication safety in the emergency department. Ann Emerg Med 2018;71:e17-e24.
- Ghaleb MA, Barber N, Franklin BD, Yeung VW, Khaki ZF, Wong IC. Systematic review of medication errors in pediatric patients. Ann Pharmacother. 2006;40:10:1766-1776.
- Gummin DD, Mowry JB, Beuhler MC, et.al. 2019 Annual Report of the American Association of Poison Control Centers’ National Poison Data System (NPDS): 37th Annual Report. Clin Tox 58:12:1360-1541.
- Panagiot M, Khan K, Keers Rn, et.al. Prevalence, severity and nature of preventable patient harm across medical care settings: systematic review and meta-analysis. BMJ. 2019:366-1418S. Available at: https://www.bmj.com/content/bmj/366/bmj.l4185.full.pdf. Accessed December 29, 2020.
- Medication use and spending in the U.S.: a review of 2018 and outlook to 2023. IQVIA Institute. May 2019. Available at: https://www.iqvia.com/insights/the-iqvia-institute/reports/the-global-use-of-medicine-in-2019-and-outlook-to-2023. Accessed December 29, 2020.
- Flynn EA, Barker KN, Carnahan BJ. National observational study of prescription dispensing accuracy and safety in 50 pharmacies. J Am Pharm Assoc. 2003;43:2:191-200.
- Campbell PJ, Patel M, Martin JR, et al. Systematic review and meta-analysis of community pharmacy error rates in the USA: 1993-2015. BMJ Open Qual. 2018;7:4:e000193.
- Institute of Medicine, Committee on Identifying and Preventing Medication Errors, Preventing Medication Errors. National Academies Press; 2007:124-125. Available at: http://nap.edu/11623. Accessed December 28, 2020.
- Grissinger MG, Globus NJ, Fricker MP. The role of managed care pharmacy in reducing medication errors. JMCP 2003;9:1:62-65.
- About medication errors. National Coordinating Council for Medication Error and Prevention website. Available at: https://www.nccmerp.org/about-medication-errors. Accessed December 28, 2020.
- Hayward RA, Asch SM, Hogan MM, Hofer TP, Kerr EA. Sins of omission: getting too little medical care may be the greatest threat to patient safety. J Gen Intern Med. 2005;20:8:686-691.
- Key element of the medication-use system. Institute for Safe Medication Practices website. Available at: https://www.ismp.org/ten-key-elements. Accessed December 28, 2020.
- Chidambaran V, Sadhasivam S, Mahmoud M. Codeine and opioid metabolism: implications and alternatives for pediatric pain management. Curr Opin Anaesthesiol. 2017;30:3:329-56.
- Food & Drug Administration. FDA Drug Safety Communication: FDA requires labeling changes for prescription opioid cough and cold medicines to limit their use to adults 18 years and older. https://www.fda.gov/Drugs/DrugSafety/ucm590435.htm. January 22, 2018. Accessed January 9, 2021.
- Eiland LS, Benner K, Gumpper KF, et. al. ASHP-PPAG guidelines for providing pediatric pharmacy services in hospitals and health systems. Am J Health Syst Pharm. 2018;75:15:1151-1165.
- Chai G, Governale L, McMahon AE, et.al. Trends of outpatient prescription drug utilization in us children, 2002–2010. Pediatrics 2012;130 :1:23-31.
- Hales CM, Kit BK, Gu Q, Ogden CL. Trends in prescription medication use among children and adolescents—United States, 1999-2014. JAMA. 2018;319:19:2009–2020.
- New pediatric labeling database. U.S. Food and Drug Administration website. August 31, 2020. Available at: https://www.accessdata.fda.gov/scripts/sda/sdNavigation.cfm?sd=labelingdatabase. Accessed December 29, 2020.
- Pharmacist liability claim report: 2nd edition. Identifying and addressing professional liability exposures. CAN/Health Professional Services Organization. 2019. Available at: http://www.hpso.com/Documents/Risk%20Education/individuals/Claim-Reports/Pharmacist/HPSO-CNA-Pharmacist-Claim-Report-2019.pdf. Accessed December 29, 2020.
- Results of pediatric medication safety survey (part 2): comparing data subsets points out areas for improvement. Institute for Safe Medication Practices website. July 2, 2015. Available at: https://www.ismp.org/resources/results-pediatric-medication-safety-survey-part-2-comparing-data-subsets-points-out-areas. Accessed January 4, 2021.
- Rinke ML, Bundy DG, Velasquez CA, et. al. Interventions to reduce pediatric medication errors: a systematic review. Pediatrics 2014;134:2:338-360.
- Ammenwerth E, Schnell-Inderst P, Machan C, Siebert U. The effect of electronic prescribing on medication errors and adverse drug events: a systematic review. J Am Med Inform Assoc. 2008;15:5:585-600.
- Porterfield A, Engelbert K, Coustasse A. Electronic prescribing: improving the efficiency and accuracy of prescribing in the ambulatory care setting. Perspect Health Inf Manag. 2014;11(Spring):1g.
- Recommendations to reduce medication errors associated with verbal medication orders and prescriptions. May 1, 2015. National Coordinating Council for Medication Error Reporting and Prevention website. Available at: https://www.nccmerp.org/recommendations-reduce-medication-errors-associated-verbal-medication-orders-and-prescriptions. Accessed December 29, 2020.
- Official “do not use” list. March 5, 2009. The Joint Commission website. Available at: https://www.jointcommission.org/-/media/deprecated-unorganized/imported-assets/tjc/system-folders/topics-library/dnu_listpdf.pdf?db=web&hash=6308831BDDB4BA3F046BB995E868DE2D. Accessed December 7, 2020.
- The patient care process for delivering comprehensive medication management (CMM): optimizing medication use in patient-centered, team-based care settings. CMM in Primary Care Research Team. July 2018. Available at http://www.accp.com/cmm_care_process. Accessed January 11, 2021.
- Lavan AH, Gallagher PF, O'Mahony D. Methods to reduce prescribing errors in elderly patients with multimorbidity. Clin Interv Aging. 2016;11:857-866.
- FDA and ISMP lists of look-alike drug names with recommended tall man letters. 2016. Institute for Safe Medication Practices website. Available at: https://www.ismp.org/sites/default/files/attachments/2017-11/tallmanletters.pdf. Accessed January 12, 2021.
- ISMP list of high-alert medications in community/ambulatory healthcare. 2011. Institute for Safe Medication Practices Website. Available at:https://www.ismp.org/recommendations/high-alert-medications-community-ambulatory-list. Accessed January 28, 2021.
- Maaskant JM, Eskes A, van Rijn-Bikker P, et. al. High-alert medications for pediatric patients: an international modified Delphi study. Expert Opin Drug Saf. 2013;12:6:805-14.
- Neuspiel DR, Taylor MM. Reducing the risk of harm from medication errors in children. Health Services Insights. January 2013. doi:10.4137/HSI.S10454
- Yin HS, Mendelsohn AL, Wolf MS, et al. Parents' medication administration errors: role of dosing instruments and health literacy. Arch Pediatr Adolesc Med. 2010;164:2:181–186.
- Dosage delivery devices for OTC liquid drug products. Food and Drug Administration Web site. Available at: https://www.fda.gov/media/78087/download. Accessed January 12, 2021.
- Budnitz DS, Lovegrove MC, Rose KO. Adherence to label and device recommendations for over-the-counter pediatric liquid medications. Pediatrics. 2014;133:2:e283-90.
- Yin HS, Mendelsohn AL, Fierman A, van Schaick L, Bazan IS, Dreyer BP. Use of a pictographic diagram to decrease parent dosing errors with infant acetaminophen: a health literacy perspective. Acad Pediatr. 2011;11:50–57.
- Safety check: put your medicines up and away and out of sight. Institute for Safe Medication Practices. September 14, 2012. ConsumerMedSafety.org website. Accessed January 11, 2021.
- Lam N, Muravez SN, Boyce RW. A comparison of the Indian Health Service counseling technique with traditional, lecture-style counseling. J Am Pharm Assoc (2003). 2015;55:503-510.
- Error reporting and analysis. Institute for Safe Medication Practices website. Available at: https://www.ismp.org/error-reporting-programs. Accessed January 12, 2021.
Back to Top